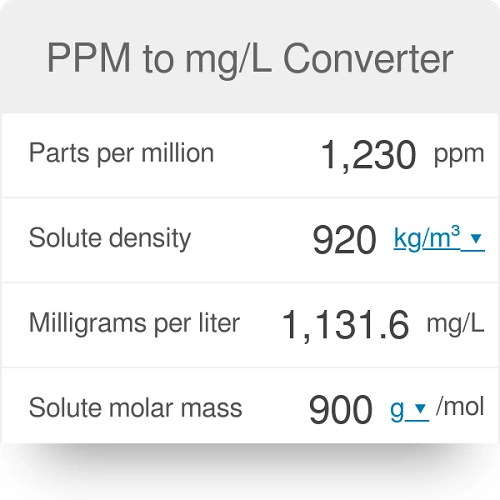
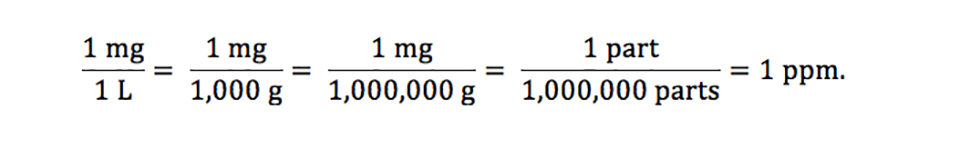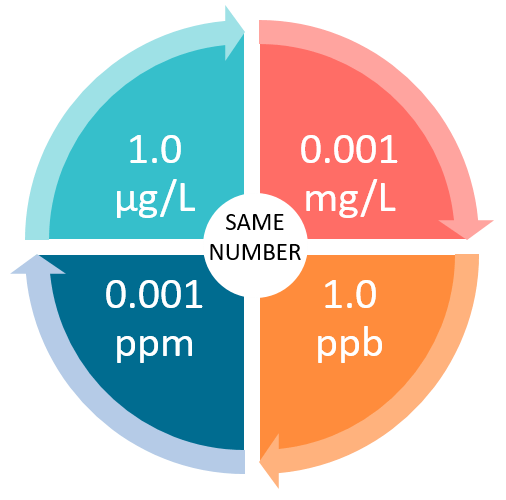OneClass: a) 1 mg/mL = ______ ppb and ______ ppm b) 1 ng/L = ______ ppb and ______ ppm c) 1 g/kg = ...

Concentrations expressed as mg L -1 (ppm) by labs must be divided by... | Download Scientific Diagram

SOLVED: A 190-mg sample of Na2CO3 is dissolved in H2O to give 1.00L of solution. What is the concentration of Na+ in parts per million (ppm)? Note: "ppm" are 106 ×[(mass of