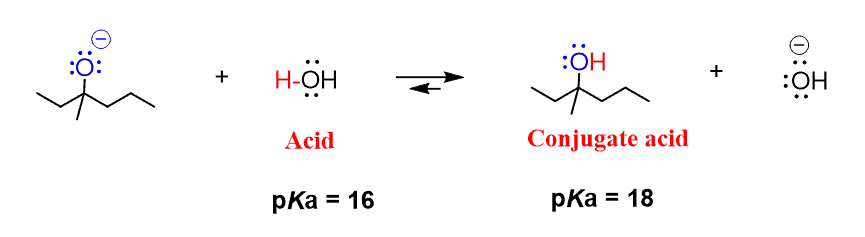
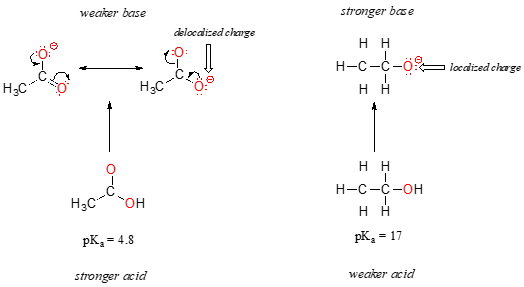
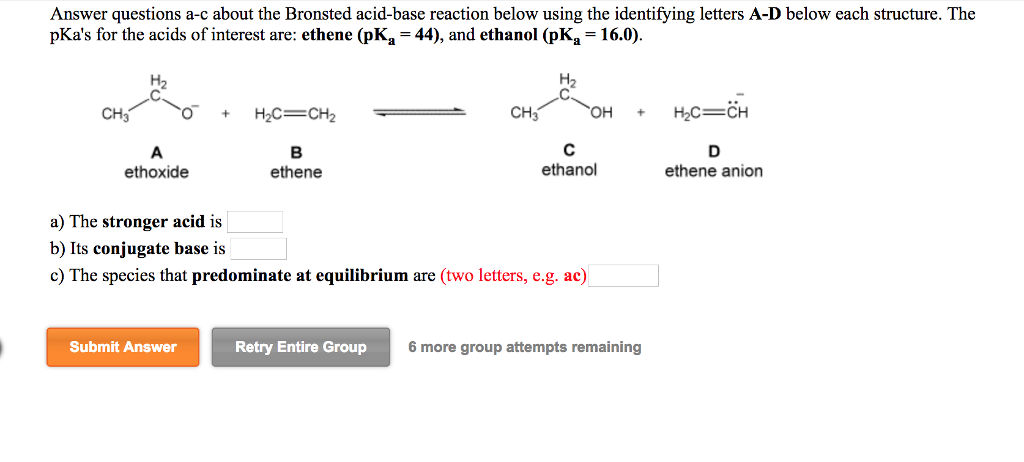
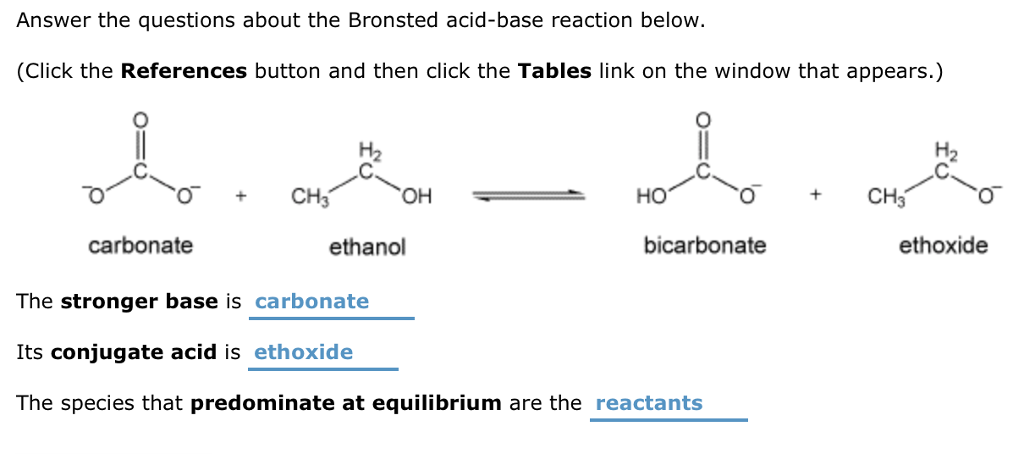
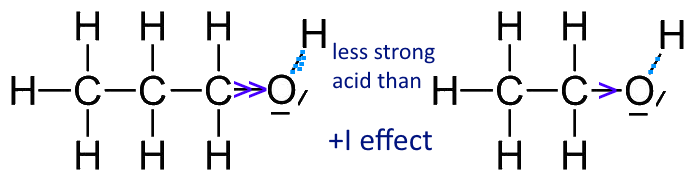
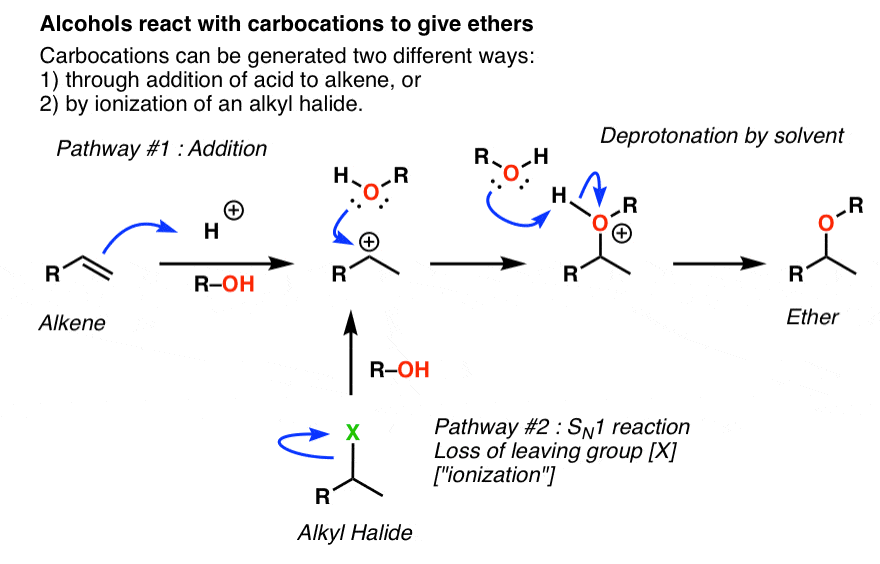
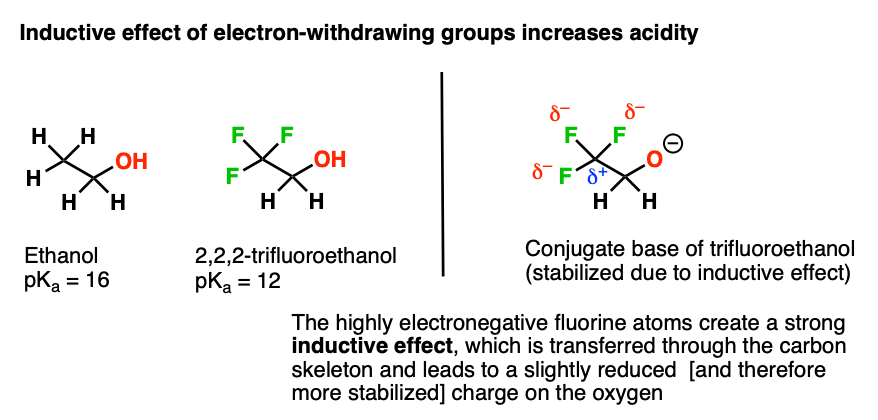
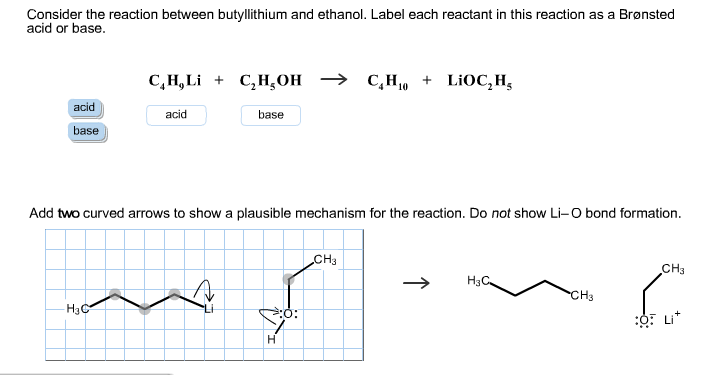
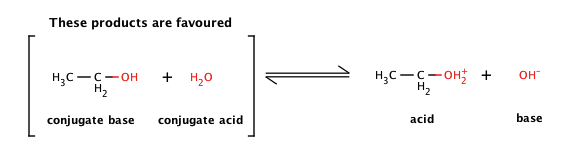
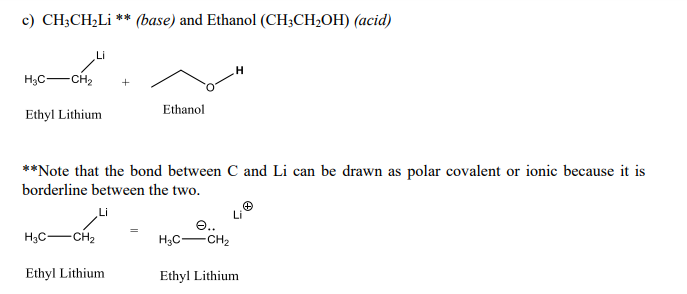
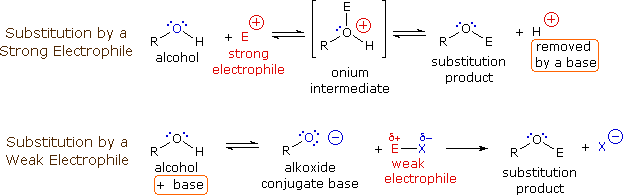
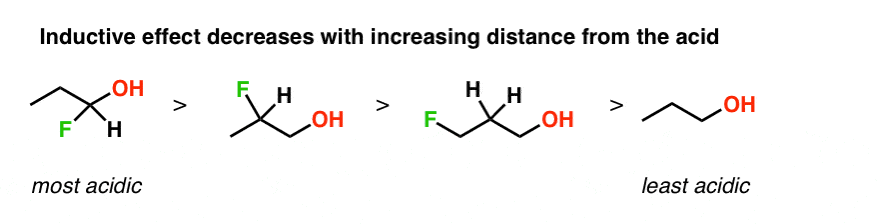
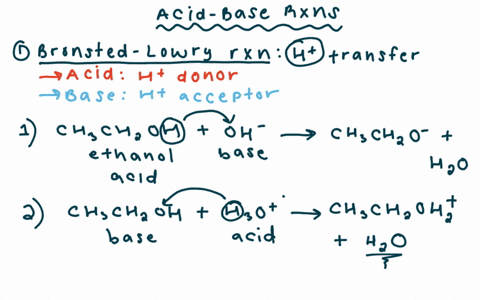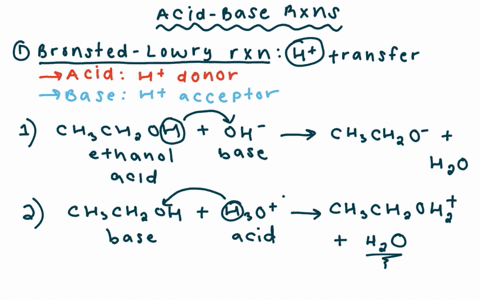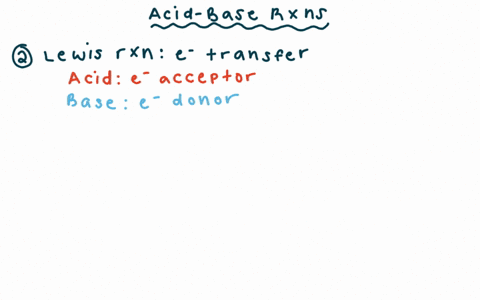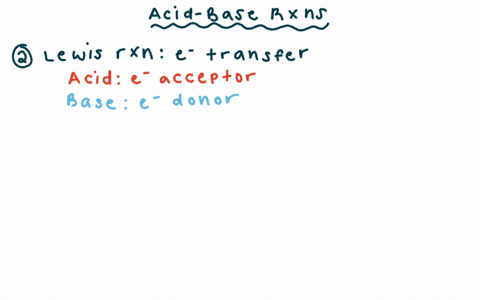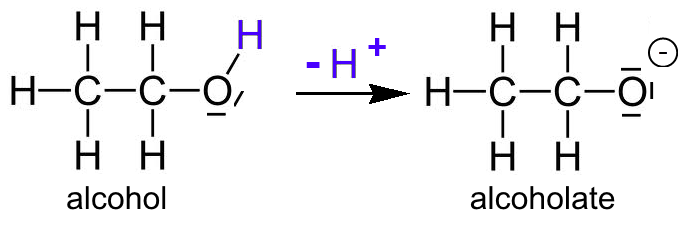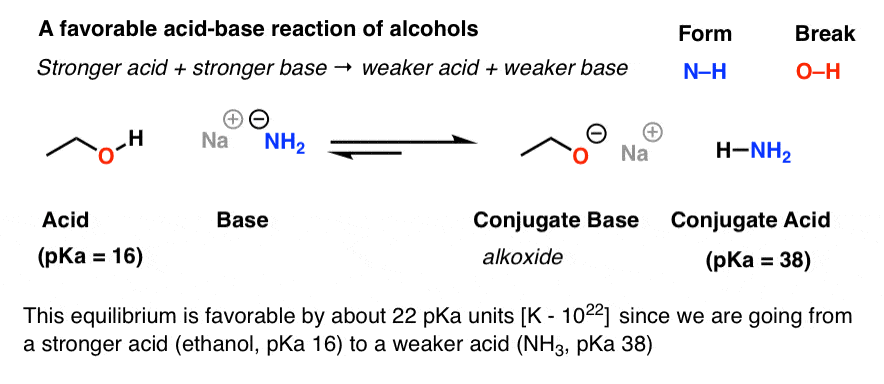Draw the Lewis structure for the conjugate base from the reaction of ethanol with a generic base. Include all lone pairs of electrons and any nonzero formal charges. | Homework.Study.com

Influence of acid–base properties on the Lebedev ethanol-to-butadiene process catalyzed by SiO2–MgO materials - Catalysis Science & Technology (RSC Publishing)

Key Roles of Lewis Acid–Base Pairs on ZnxZryOz in Direct Ethanol/Acetone to Isobutene Conversion | Journal of the American Chemical Society